Modern Energy Sources and Challenges
Increasing population is increasing energy demand therefore putting additional pressure on the energy sector using fossil fuels for energy generation. Moreover, finite supply of fossil fuels and increasing negative environmental impact is pushing us to seek an alternative source of energy, that is where the use of renewable energy sector is blooming. By now many developed nations have implemented major efforts in the renewable energy sector, reducing overall use of fossil fuels. There are few renewable energy sources that are attracting more interest such as solar power, wind power, biomass energy, hydropower, geothermal energy and hydrogen fuel, among which hydrogen has been identified as the cleanest option, due to zero carbon emissions processes compared to conventional fossil fuels. However major challenge in H2 is its cost intensive storage and transportation. H2 can be stored as gas, liquid or solid. For instance, compressing gas requires a pressure of about 700 bar and storing as liquid requires cryogenic condition for cooling down to -253˚C [1]. Therefore, storing compressed gas or liquid is cost intensive process also requiring expensive infrastructure. H2 can be stored as solid either physically or chemically, generally more H2 can be stored in same volume as the solid H2 storage in same volume compared to liquid and gas. During physical storage, H2 can be stored in microporous or tubular structures, at ambient condition, the most commonly use solid H2 carriers are metal organic frameworks (MOF). During chemical storage, H2 can be chemically bonded in molecules like metal borohydride, metal aluminum hydride, and some amides and imides. In these materials, H2 gas adsorption is an exothermic process, therefore it releases heat during storing of H2, this requires some sophisticated infrastructure to dissipate the heat, making an overall costly infrastructure and proper heat management. Moreover, the adsorbing H2 is a kinetically slow process, potentially irreversible, requires high dehydrogenation temperature and hence costly process [2].
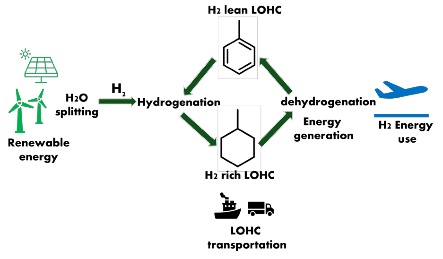
Figure 1: Schematic of liquid organic hydrogen carriers (LOHC) from production, H2 storage, H2 release, utilization and cycling.
Liquid organic hydrogen carrier
Liquid organic hydrogen carrier (LOHC) is another type of hydrogen storage materials through a reversible catalytic hydrogenation of unsaturated molecules such as toluene, methylcyclohexane (MCH), N-ethylcarbazole, perhydro-dibenzyl toluene (H18-DBT), storing H2 as liquid in ambient condition. LOHC attracted attention due to its safety and practicality and use of existing infrastructure for liquid fuels. However, one of the significant limitations of LOHC is requiring high enthalpy for releasing H2, requiring high temperature, reduces the efficiency of hydrogen release from LOHC. Therefore, there still lies a challenge for improving conventional LOHC.
| Compressed H2 | Liquified H2 | Metal hydride | Adsorbents/MOF | LOHC | |
| H2 content (wt% H2) | 100 | 100 | 1-8 | 1-10 | 6-7 |
| Volumetric density gH2/L | 23-61 | 71.7 | 50-150 | 20-50 | 40-70 |
| Safety | Low due to high pressure use | Low due to boil off explosion risk | Low due to some pyrophoric nature | Good/inert | Varies with respect to the molecule |
| Infrastructure use | Established tech, fast refueling | Need cryogenic conditions | Complex | New technology needed | Can use existing infrastructure |
A few necessary properties of the LOHC are high hydrogen storage (>5 wt% H2), reversible hydrogenation and dehydrogenation, thermodynamic favorability at moderate temperature and pressure and fast kinetics of hydrogenation-dehydrogenation by catalyst, a suitable enthalpy for dehydrogenation. In addition, chemical stability of the storage material, safer handing and lower toxicity are few core requirements of the LOHC. Nature of catalyst also plays a key role in the hydrogenation and hydrogenation cycle, that needs to be properly studied.
Can AI Help Identify Efficient LOHCs
AI is a futuristic tool that can be used for material discovery, to identify a good LOHC. For instance, a team at Argonne National Laboratory [3] used AI to screen 160 billion molecules for LOHC, narrowing down to 41 promising candidates (figure 2). In this study consisted of similarity of known LOHCs, physical properties, hydrogen storage capacity and dehydrogenation enthalpy to screen the molecules. The AI model developed for this system analyzed 3 million molecules per second, therefore completing the whole screening in 14 hours that would otherwise take 5 years. Therefore, AI is going to be a core tool for future technology innovation, to use in molecule discovery, accelerating the innovation. Figure 2 represents a series of overlapping spaces. The largest cycle here represents the vast chemical compound space of ̴1011 molecules (candidates for LOHC), the smaller circle within represents the unsaturated molecule. The four small circles inside represent the additional filters on structure (similarity of known LOHC), practicality (synthesizability and favorable physical properties, capacity (a gravimetric hydrogen capacity of ≥5.5%), and enthalpy (of dehydrogenation between 40-70 kJ/mol of H2). The intersection of these four circles highlights the most promising candidates for LOHC.
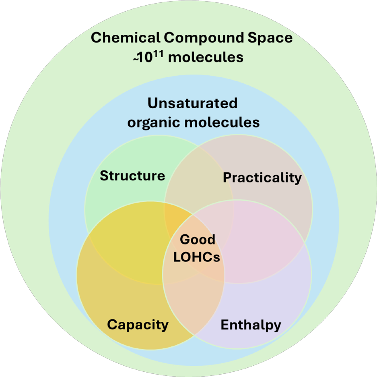
Figure 2: Visual representation of the series of nested and overlapping spaces used to predict the good LOHC using AI model. Figure is adapted from reference [3].
Conclusion
Overall, for practical applications of LOHC, there are several factors to consider are, thermodynamics, kinetics, reversibility of the H2 storage and release, safety, cost and overall efficiency of the process. In addition, there is a huge scope in the future to advance AI models to take more parameters into account such as catalyst (metal types), catalyst size/shape to advance the materials screening and material discovery for a promising LOHC.
Reference
- Hydrogen and Fuel Cell Technologies Office
- Kim, H.; So, S. H.; Muhammad, R.; Oh, H. Comparing the Practical Hydrogen Storage Capacity of Porous Adsorbents: Activated Carbon and Metal‑Organic Framework. Int. J. Hydrogen Energy. 2024, 50, 1616–1625.
- Argonne National Laboratory. Argonne Researchers Use AI to Downselect 160 Billion Molecules to Just 41 Candidates for Hydrogen Liquid Carriers. Green Car Congress. 2024.

Author
